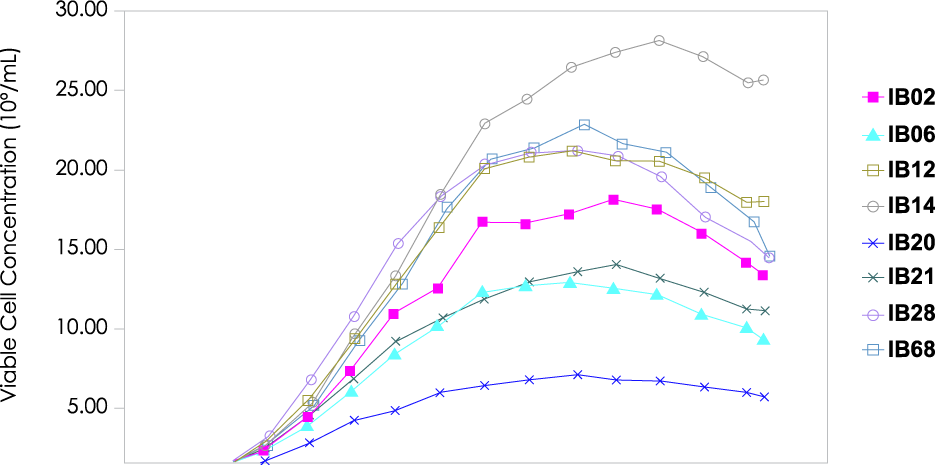
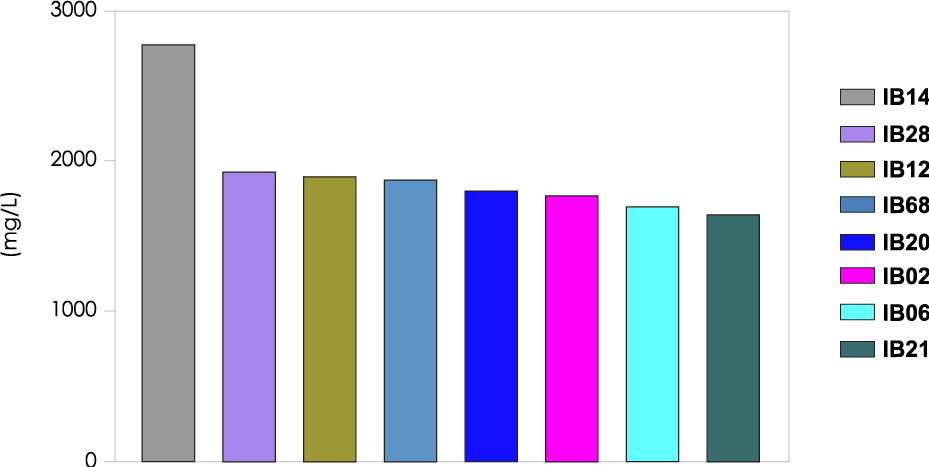
Solid Tumors and Unmet Needs
Advances in the field of immuno-oncology have led to new and better treatment outcomes for a range of cancers, and particularly, blood cancers. However, even with the advent of checkpoint inhibitors as immunotherapies for solid tumors, significant challenges remain. This is in part due to dynamics in the tumor microenvironment, wherein regulatory T cells [Tregs] proliferate and suppress the immune responses to tumor cells.
The Treg is a type of T cell that is important in preventing autoimmunity by keeping effector T cells [Teffs] “in-check” to prevent destruction of healthy tissues. However, around tumors, Tregs create an immunosuppressive environment and block the work of Teffs to do what they do well – kill cancer cells. So, targeting depletion of Tregs to control tumors has emerged as an area of interest in oncology over the past several years.
Fortunately, Tregs express a protein, CD25, that allows them to be killed with an anti-CD25 antibody. Unfortunately, that protein is part of the interleukin-2 [IL-2] signaling, which also activates Teffs. This is what has presented a challenge; anti-CD25 antibodies can deplete Tregs, but unless they have certain characteristics, those antibodies also prevent Teffs from cancer cell killing, thereby resulting in a failure as a therapeutic to control tumor growth and metastasis.